What Is The Chemical Makeup Of Lipids
7.3: Lipids
- Page ID
- 5308
Learning Objectives
- Describe the chemical composition of lipids
- Describe the unique characteristics and diverse structures of lipids
- Compare and dissimilarity triacylglycerides (triglycerides) and phospholipids
- Describe how phospholipids are used to construct biological membranes
Although they are composed primarily of carbon and hydrogen, lipid molecules may too contain oxygen, nitrogen, sulfur, and phosphorous. Lipids serve numerous and diverse purposes in the construction and functions of organisms. They tin be a source of nutrients, a storage form for carbon, energy-storage molecules, or structural components of membranes and hormones. Lipids comprise a broad class of many chemically distinct compounds, the most common of which are discussed in this department.
Fatty Acids and Triacylglycerides
The fatty acids are lipids that contain long-chain hydrocarbons terminated with a carboxylic acid functional group. Because of the long hydrocarbon concatenation, fat acids are hydrophobic ("water fearing") or nonpolar. Fatty acids with hydrocarbon chains that comprise only single bonds are called saturated fatty acids because they accept the greatest number of hydrogen atoms possible and are, therefore, "saturated" with hydrogen. Fatty acids with hydrocarbon chains containing at least one double bond are called unsaturated fatty acids because they accept fewer hydrogen atoms. Saturated fatty acids have a straight, flexible carbon backbone, whereas unsaturated fatty acids have "kinks" in their carbon skeleton considering each double bond causes a rigid curve of the carbon skeleton. These differences in saturated versus unsaturated fatty acid structure result in different properties for the corresponding lipids in which the fat acids are incorporated. For case, lipids containing saturated fatty acids are solids at room temperature, whereas lipids containing unsaturated fatty acids are liquids.
A triacylglycerol, or triglyceride, is formed when 3 fatty acids are chemically linked to a glycerol molecule (Figure \(\PageIndex{1}\)). Triglycerides are the master components of adipose tissue (body fat), and are major constituents of sebum (peel oils). They play an important metabolic office, serving as efficient energy-storage molecules that can provide more than than double the caloric content of both carbohydrates and proteins.
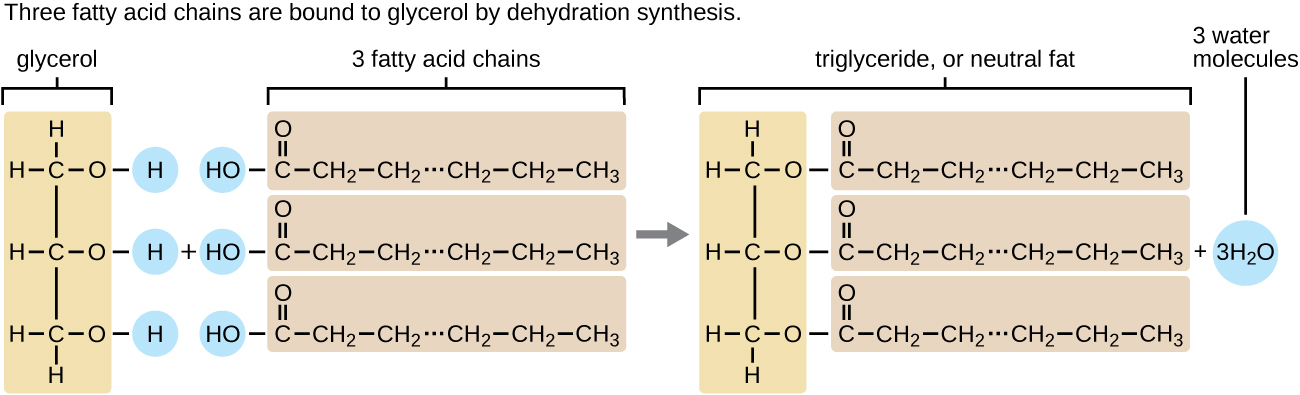
Practise \(\PageIndex{1}\)
Explain why fat acids with hydrocarbon chains that comprise simply single bonds are called saturated fatty acids.
Phospholipids and Biological Membranes
Triglycerides are classified as simple lipids because they are formed from just ii types of compounds: glycerol and fatty acids. In dissimilarity, complex lipids contain at to the lowest degree 1 boosted component, for example, a phosphate group (phospholipids) or a carbohydrate moiety (glycolipids). Effigy \(\PageIndex{2}\) depicts a typical phospholipid composed of two fatty acids linked to glycerol (a diglyceride). The 2 fat acid carbon chains may be both saturated, both unsaturated, or one of each. Instead of some other fatty acid molecule (every bit for triglycerides), the third binding position on the glycerol molecule is occupied by a modified phosphate group.
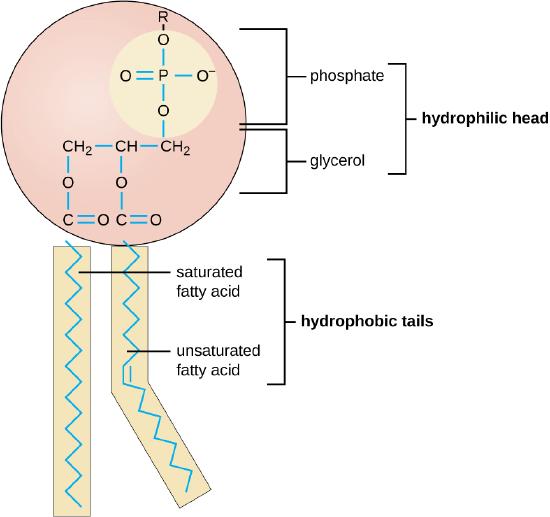
The molecular construction of lipids results in unique behavior in aqueous environments. Figure \(\PageIndex{1}\) depicts the structure of a triglyceride. Because all iii substituents on the glycerol backbone are long hydrocarbon chains, these compounds are nonpolar and not significantly attracted to polar water molecules—they are hydrophobic. Conversely, phospholipids such as the one shown in Figure \(\PageIndex{2}\) have a negatively charged phosphate group. Because the phosphate is charged, it is capable of strong attraction to water molecules and thus is hydrophilic, or "h2o loving." The hydrophilic portion of the phospholipid is oftentimes referred to every bit a polar "caput," and the long hydrocarbon chains as nonpolar "tails." A molecule presenting a hydrophobic portion and a hydrophilic moiety is said to be amphipathic. Detect the "R" designation within the hydrophilic head depicted in Figure \(\PageIndex{2}\), indicating that a polar head grouping can be more complex than a simple phosphate moiety. Glycolipids are examples in which carbohydrates are bonded to the lipids' head groups.
The amphipathic nature of phospholipids enables them to form uniquely functional structures in aqueous environments. As mentioned, the polar heads of these molecules are strongly attracted to water molecules, and the nonpolar tails are non. Because of their considerable lengths, these tails are, in fact, strongly attracted to one another. Every bit a result, energetically stable, big-scale assemblies of phospholipid molecules are formed in which the hydrophobic tails congregate within enclosed regions, shielded from contact with h2o by the polar heads (Figure \(\PageIndex{3}\)). The simplest of these structures are micelles, spherical assemblies containing a hydrophobic interior of phospholipid tails and an outer surface of polar caput groups. Larger and more complex structures are created from lipid-bilayer sheets, or unit membranes, which are large, two-dimensional assemblies of phospholipids congregated tail to tail. The cell membranes of almost all organisms are made from lipid-bilayer sheets, equally are the membranes of many intracellular components. These sheets may as well grade lipid-bilayer spheres that are the structural basis of vesicles and liposomes, subcellular components that play a role in numerous physiological functions.
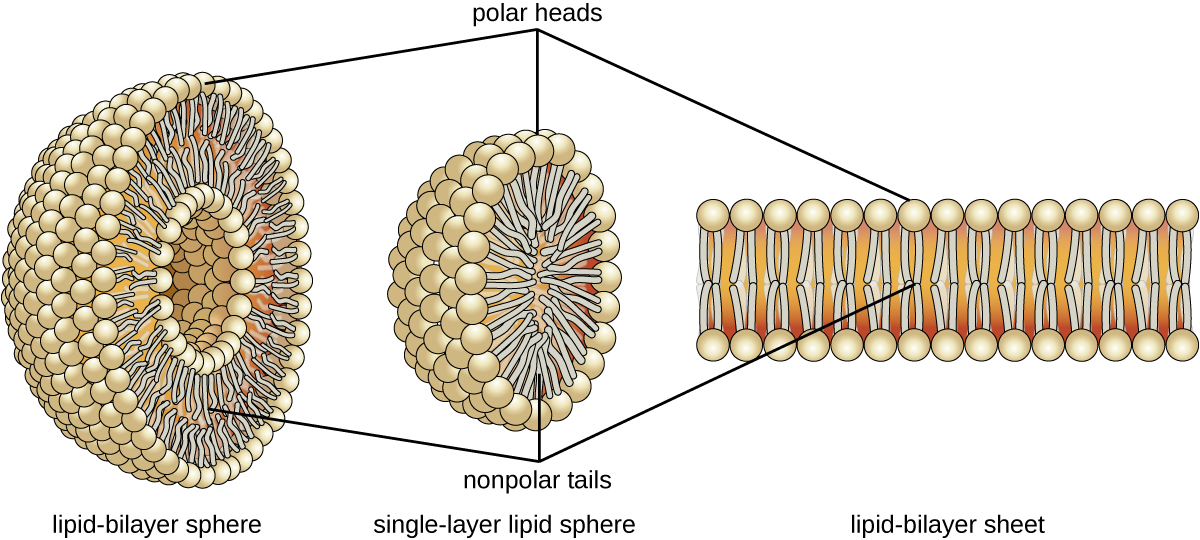
Exercise \(\PageIndex{2}\)
How is the amphipathic nature of phospholipids meaning?
Isoprenoids and Sterols
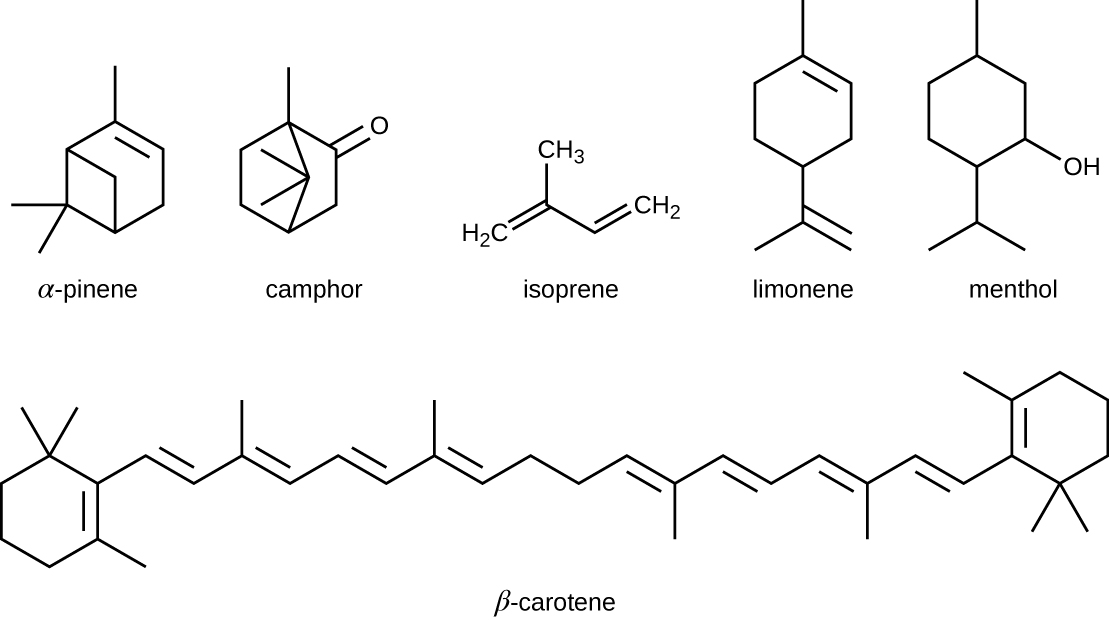
The isoprenoids are branched lipids, also referred to as terpenoids, that are formed by chemical modifications of the isoprene molecule (Effigy \(\PageIndex{4}\)). These lipids play a broad variety of physiological roles in plants and animals, with many technological uses as pharmaceuticals (capsaicin), pigments (e.g., orange beta carotene, xanthophylls), and fragrances (east.g., menthol, camphor, limonene [lemon fragrance], and pinene [pine fragrance]). Long-concatenation isoprenoids are besides institute in hydrophobic oils and waxes. Waxes are typically h2o resistant and hard at room temperature, only they soften when heated and liquefy if warmed adequately. In humans, the main wax production occurs within the sebaceous glands of hair follicles in the skin, resulting in a secreted material chosen sebum, which consists mainly of triacylglycerol, wax esters, and the hydrocarbon squalene. In that location are many leaner in the microbiota on the skin that feed on these lipids. Ane of the well-nigh prominent bacteria that feed on lipids is Propionibacterium acnes, which uses the skin's lipids to generate short-chain fat acids and is involved in the production of acne.
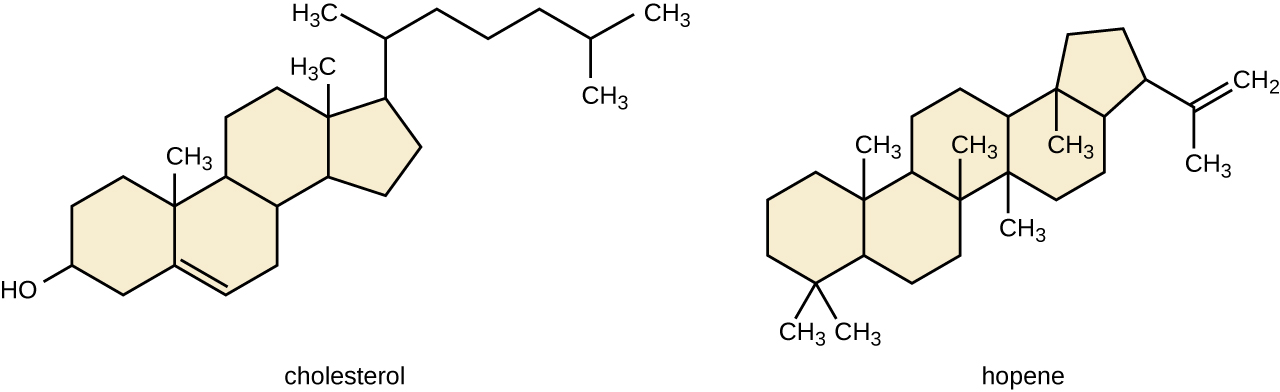
Another blazon of lipids are steroids, complex, ringed structures that are institute in prison cell membranes; some function equally hormones. The nigh common types of steroids are sterols, which are steroids containing an OH group. These are mainly hydrophobic molecules, just also have hydrophilic hydroxyl groups. The well-nigh common sterol establish in animal tissues is cholesterol. Its structure consists of four rings with a double bond in one of the rings, and a hydroxyl group at the sterol-defining position. The office of cholesterol is to strengthen prison cell membranes in eukaryotes and in bacteria without cell walls, such every bit Mycoplasma. Prokaryotes more often than not do non produce cholesterol, although bacteria produce like compounds chosen hopanoids, which are likewise multiringed structures that strengthen bacterial membranes (Effigy \(\PageIndex{v}\)). Fungi and some protozoa produce a similar compound called ergosterol, which strengthens the jail cell membranes of these organisms.
This video provides boosted information most phospholipids and liposomes.
Exercise \(\PageIndex{3}\)
How are isoprenoids used in applied science?
Clinical Focus: Part 2
The moisturizing foam prescribed by Penny'due south doctor was a topical corticosteroid cream containing hydrocortisone. Hydrocortisone is a constructed form of cortisol, a corticosteroid hormone produced in the adrenal glands, from cholesterol. When applied straight to the peel, it tin can reduce inflammation and temporarily relieve pocket-size skin irritations, itching, and rashes by reducing the secretion of histamine, a chemical compound produced by cells of the immune organization in response to the presence of pathogens or other foreign substances. Considering histamine triggers the body'due south inflammatory response, the ability of hydrocortisone to reduce the local production of histamine in the skin effectively suppresses the immune system and helps limit inflammation and accompanying symptoms such as pruritus (itching) and rashes.
Practise \(\PageIndex{4}\)
Does the corticosteroid cream treat the cause of Penny's rash, or but the symptoms?
Central Concepts and Summary
- Lipids are equanimous mainly of carbon and hydrogen, simply they can also incorporate oxygen, nitrogen, sulfur, and phosphorous. They provide nutrients for organisms, store carbon and energy, play structural roles in membranes, and office every bit hormones, pharmaceuticals, fragrances, and pigments.
- Fat acids are long-concatenation hydrocarbons with a carboxylic acrid functional group. Their relatively long nonpolar hydrocarbon bondage make them hydrophobic. Fatty acids with no double bonds are saturated; those with double bonds are unsaturated.
- Fatty acids chemically bail to glycerol to course structurally essential lipids such every bit triglycerides and phospholipids. Triglycerides comprise three fatty acids bonded to glycerol, yielding a hydrophobic molecule. Phospholipids contain both hydrophobic hydrocarbon bondage and polar head groups, making them amphipathic and capable of forming uniquely functional large scale structures.
- Biological membranes are large-scale structures based on phospholipid bilayers that provide hydrophilic outside and interior surfaces suitable for aqueous environments, separated by an intervening hydrophobic layer. These bilayers are the structural ground for jail cell membranes in most organisms, too as subcellular components such as vesicles.
- Isoprenoids are lipids derived from isoprene molecules that have many physiological roles and a multifariousness of commercial applications.
- A wax is a long-chain isoprenoid that is typically water resistant; an case of a wax-containing substance is sebum, produced by sebaceous glands in the peel. Steroids are lipids with complex, ringed structures that function as structural components of jail cell membranes and as hormones. Sterols are a subclass of steroids containing a hydroxyl group at a specific location on one of the molecule'southward rings; ane example is cholesterol.
- Bacteria produce hopanoids, structurally like to cholesterol, to strengthen bacterial membranes. Fungi and protozoa produce a strengthening agent called ergosterol.
What Is The Chemical Makeup Of Lipids,
Source: https://bio.libretexts.org/Bookshelves/Microbiology/Microbiology_%28OpenStax%29/07:_Microbial_Biochemistry/7.03:_Lipids
Posted by: gutierrezloseem.blogspot.com


0 Response to "What Is The Chemical Makeup Of Lipids"
Post a Comment